The COA: a PDF You Want to Trust
Gray market peptides and the Certificate of Analysis
What Is a COA Really Measuring?
The market for research peptides is indeed a strange ecosystem. It’s a place where one slick looking internet storefront may represent a regulation-compliant supplier working in conjunction with a legitimate compounding pharmacy, whereas another near-identical one was just tossed up by a dude operating out of his bedroom in Nanjing. This market’s goods are obscure molecules with names that sound like discontinued Pokémon evolutions, and its consumers range from scientific researchers to bodybuilders who’d like to inject some complex amino chains after chugging a creatine shake.
It’s also a space rife with skepticism. There are everyday consumers grappling with a variety of health issues who’ve stumbled into the research chemical space out of skepticism towards their doctors – allopaths, osteopaths, naturopaths etc. – and the various big-money industries that seem to back them. There are scientists who’ve been watching in quiet horror as research funding has run dry.
Log onto any online forum dedicated to the discussion of peptides and you’ll find an environment florid with paranoia. People are worrying about the FDA, about the public’s waning faith in the scientific method, about doctors and their ability to treat anything beyond symptoms, about influencers and their “stacks,” about Discord channels and shady WhatsApp group buys. The list goes on.
In such an ecosystem, there are few reliable signposts of truth. Jumpy consumers feeling spurned by mainstream medicine want something else, but don’t know who to trust. Empirical data can be easily faked, as can authority; one may assume she’s speaking with an “expert” about her peptide protocol only to discover she’s been DMing an AI. Suffice to say, it’s wild out there, and a bit disheartening. If you’re reading this, you almost certainly have a sense of just how disheartening it can be.
If I were to ask how you distinguish a trustworthy peptide supplier from a scammer, what would your answer be? Something about demonstrated scientific knowledge? A personal touch instead of faceless automation? Honesty? Integrity? (Once again, the list goes on.)
“I would be most likely to trust a supplier who prioritizes research integrity and consumer safety over profits,” you might say. “Someone who’d be willing to get their peptides tested by a third-party laboratory for safety and purity.”
And you wouldn’t be wrong. To quote the founder of Peptide Partners himself: Never trust a supplier that doesn’t submit to third-party testing. Never trust a certification that cannot be independently verified.
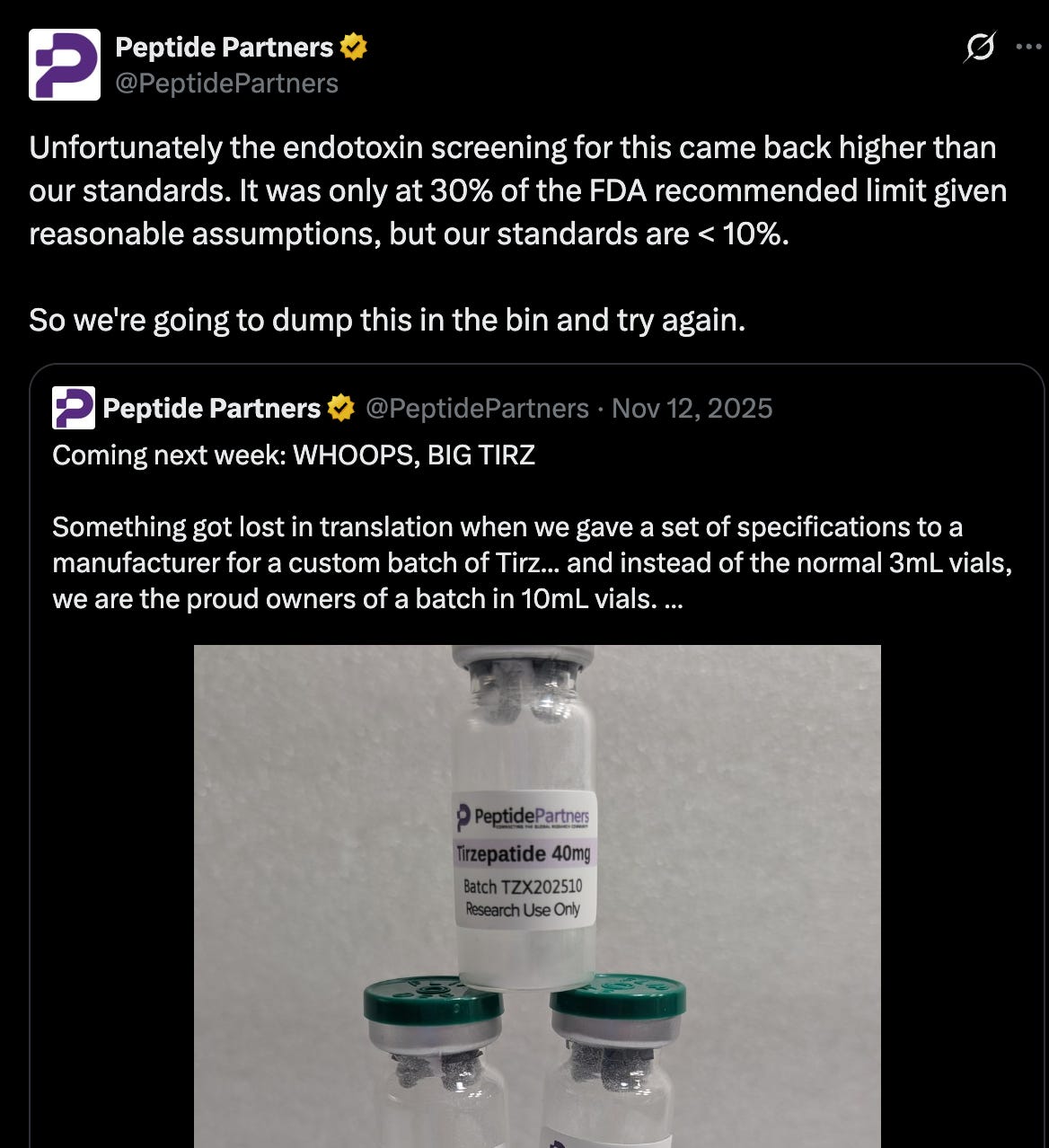
It takes a lot of humility for anyone in the rapidly expanding peptide space to pursue safety over profits – even when that means dumping hundreds of thousands of dollar’s-worth in inventory that didn’t meet quality standards.
Take a look at this compelling drama in three acts:
A batch of tirzepatide failed an endotoxin screening. Since endotoxins are extraordinarily dangerous to consumers, the company has elected to dump the batch and try again, sacrificing something in the neighborhood of $20,000 in the process. A sizable loss, and one rarely stomached in the rapid-growth peptide ecosystem. The comments reflect this:
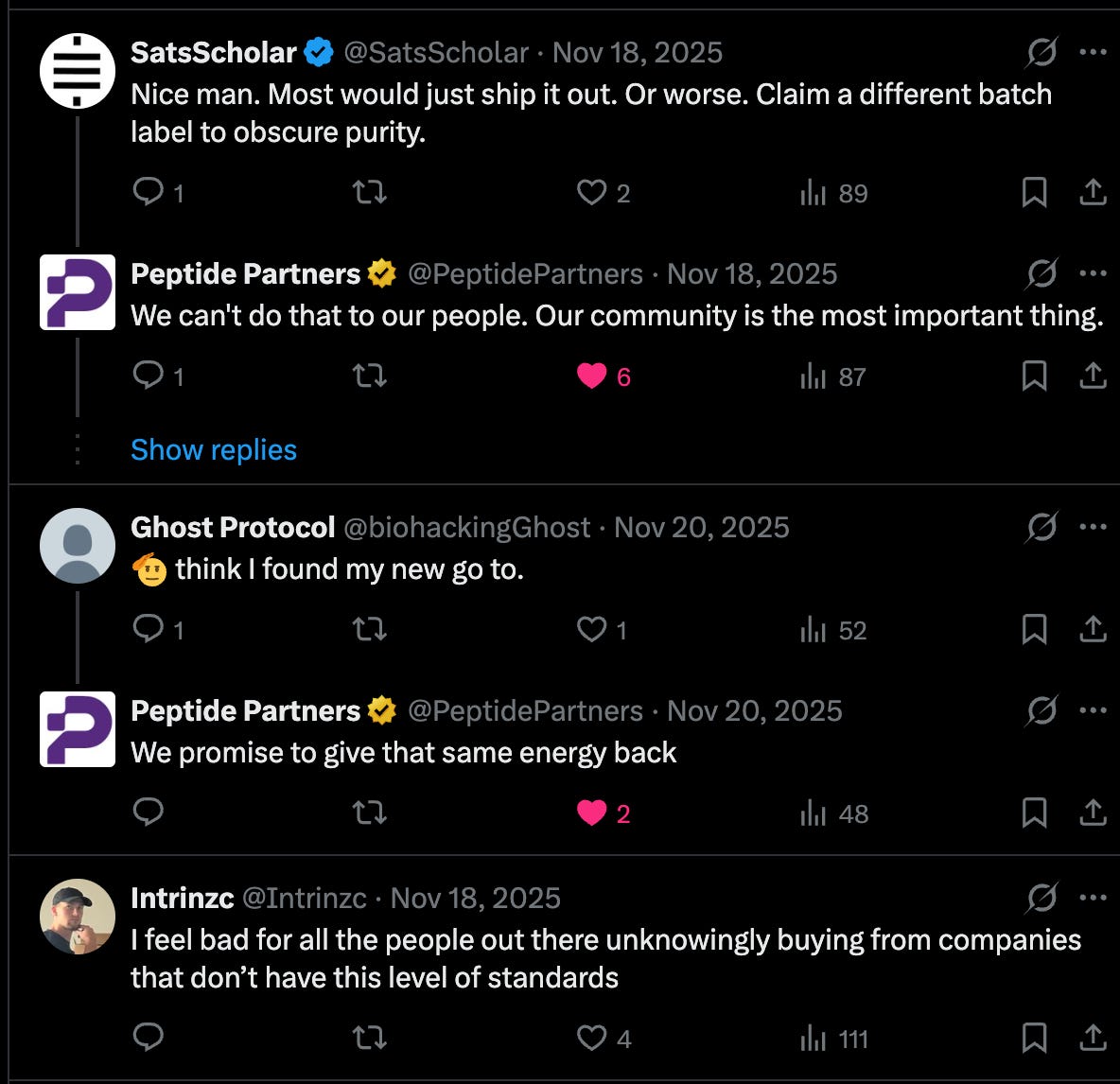
(SatsScholar has a valuable point – more on that later.)
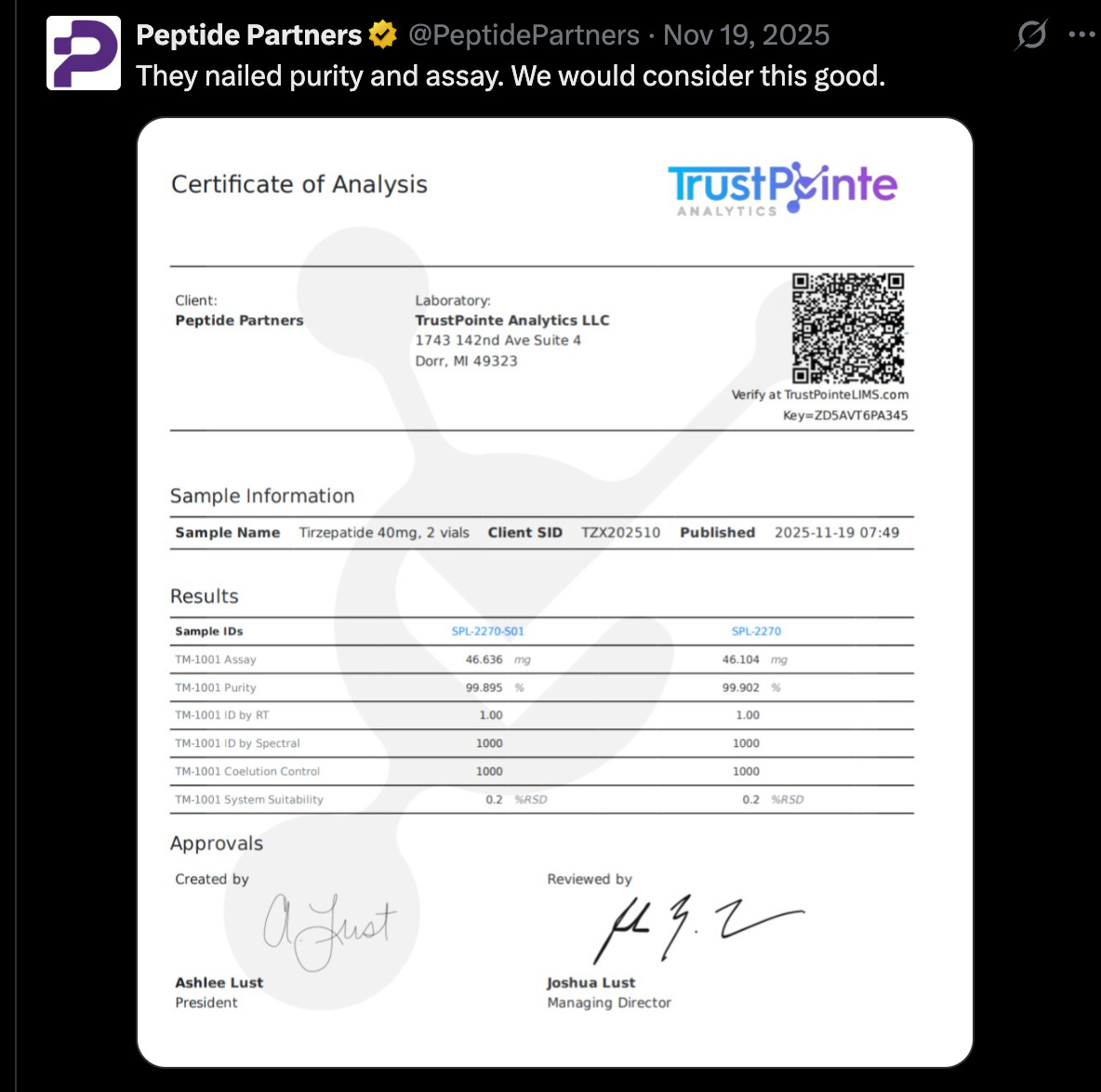
Next, the company tweets a Certificate of Analysis from TrustPointe Analytics, a chemical quality control lab specializing in peptides and other wellness products: “They nailed purity and assay. We would consider this good.”
This new batch of tirzepatide is without question a better performing one:
Here, I see just about every box checked: scientific knowledge, personal touch, honesty, and integrity. And, to paraphrase a certain transcendentalist poet known for his own eccentric wellness habits: so much depends on the COA.
So much indeed! It’s the gatekeeper, the dealbreaker, the guarantor of truth about your peptides. But what is it measuring, really?
Peptide Research, Done Cleanly — Peptide Partners. Independent HPLC/MS, batch COAs, and endotoxin screening to USP <85> validate identity and purity of peptides for research. Browse inventory and view certificates at Peptide Partners.
How the COA Works
A Certificate of Analysis is essentially a laboratory report attached to a specific batch of a compound. Its purpose is to document the results of tests that verify the identity, composition, and quality of that batch.
These tests are conducted via a variety of testing methods – e.g. mass spectrometry, gravimetric analysis, High-Performance Liquid Chromatography – designed to analyze chemical attributes such as weight, purity, and sterility.
Provided the tests go off without a hitch, the COA should inform you of three things:
Identification (Whether the substance in the vial actually is what it claims to be)
Net Content (How much of the substance is present in the vial versus non-peptide contaminants like endotoxins, heavy metals, etc.)
Net Purity (The identities of any other non-peptide substances in the vial)
Identification
The first and most fundamental anyone will want to know of the peptides they’ve just ordered: Is the thing in the vial what the label says it is?
This test is usually performed through mass spectrometry, which measures the molecular weight of the compound. Every peptide has a characteristic molecular mass determined by its amino acid sequence.
By measuring a certain peptide’s molecular mass, the Mass Spectrometer confirms its identity. For example, if the expected mass of a given peptide is 1570 daltons (a unit of molecular mass) and the Mass Spectrometer confirms 1570 daltons, that’s strong evidence that the peptide exists in the vial.
It’s important to remember that the mass spectrometry measurement is not a conclusive positive ID – other measurements must be taken as well – but it’s still a critical part of any chemical analysis.
Net Content
The next question is quantitative: How much material is actually in the vial?
Manufacturers label peptide vials with nominal quantities—5 mg, 10 mg, and so on—but those numbers are targets rather than guarantees. Net content testing determines the actual mass present in the sample through either gravimetric analysis or quantitative chromatography.
If a vial labeled 10 mg tirzepatide actually contains 8.9 mg tirzepatide, that discrepancy matters enormously. Any researcher calculating doses or concentrations from the label alone will unknowingly start their experiment with incorrect numbers. When examining a COA for net content, ensure the report explicitly states the mg per vial found, not just the purity percentage.
Net content measurements name the difference between empirical precision and guesswork.
Net Purity
Purity refers to the percentage of the material in the sample that corresponds to the intended peptide rather than synthesis byproducts, fragments, or contaminants. Purity tests are typically conducted using a method called high-performance liquid chromatography (HPLC). On a COA, it’s usually displayed as a percentage. For research purposes, you’d generally want to see net purity in excess of 98% or 99%.
Purity is also the COA metric that’s most frequently bandied about. A “high-purity” peptide sample is automatically understood to be a high-quality sample. But closer examination proves this to be a facile line of thinking:
A COA that reports 99% purity in a given peptide batch is claiming that the peptide accounts for 99% of the measured material. Reassuring, certainly, but here’s what the purity test doesn’t tell us:
Whether the remaining 1% of material is completely harmless
The chemical identities of any potential impurities
That the assay has measured everything present
Like net content and chemical ID tests, purity results are most meaningful when taken into account with other data.
Additional Quality Tests
Any third-party lab that’s serious about its chemical analyses – and by extension the lives of the individuals interacting with those chemicals – will also be sure to test for the following in any given batch of peptides:
Heavy metals
Endotoxins
Sterility
Residual solvents
These tests matter because synthesis and purification processes can introduce contaminants that can adversely affect research outcomes or biological systems. Suppliers who advertise “full panel testing” typically mean that the peptide on offer has been evaluated across several of these categories in addition to net content and net purity.
Which leads to an important point – one little discussed among the hungry-for-results gray market crowd: Not all COAs are created equal. Some contain extensive analytical data, while others contain a few numbers and a logo from a lab run by a far-flung Redditor.
Conformity Testing
Conformity testing functions as a replication check. Really, it’s the sort of simple due diligence that ought to be routine.
Instead of testing a single vial from a batch of freshly obtained Chinese peptides, the supplier chooses instead to test multiple vials from that batch. The vials are sent to a third-party lab and tested independently; the results are then compared to determine whether they align.
If the batch is consistent, the measurements across samples should fall within a very small window of variance. If not, this could indicate an uneven batch: one whose quality varies from vial to vial.
Recall the screenshots above: a high-integrity, trustworthy supplier would dump a batch that failed conformity testing – or any of the tests I’ve described – even if it meant sacrificing profits. Such a move would be anathema to a corporate machine that could care less about the human lives it’s trammeling. But for those of us in the wellness and longevity space who actually care about using science to help people get healthy, poisoning your consumer base pretty much defeats the whole point.
Strong and Weak COAs
Let’s return to SatsScholar’s comment on the Peptide Partners post about the bad batch of tirzepatide: “Nice man. Most would just ship it out. Or worse. Claim a different batch number to obscure purity.”
What does SatsScholar mean? Here, he’s referring to how easily COAs can be falsified to serve a shady supplier’s purposes. One can conveniently neglect to conformity-test a batch of peptides, or simply recycle a COA for a dubious batch and nonchalantly look the other way. And this doesn’t even begin to touch the discrepancies among labs: does Janoshik measure up to TrustPointe? Chromate to MZ Biolabs?
Without getting into the finer points of chemical assays and lab etiquette, I want to briefly outline the differences between a good – i.e. trustworthy, robust – COA and a not-so-good one.
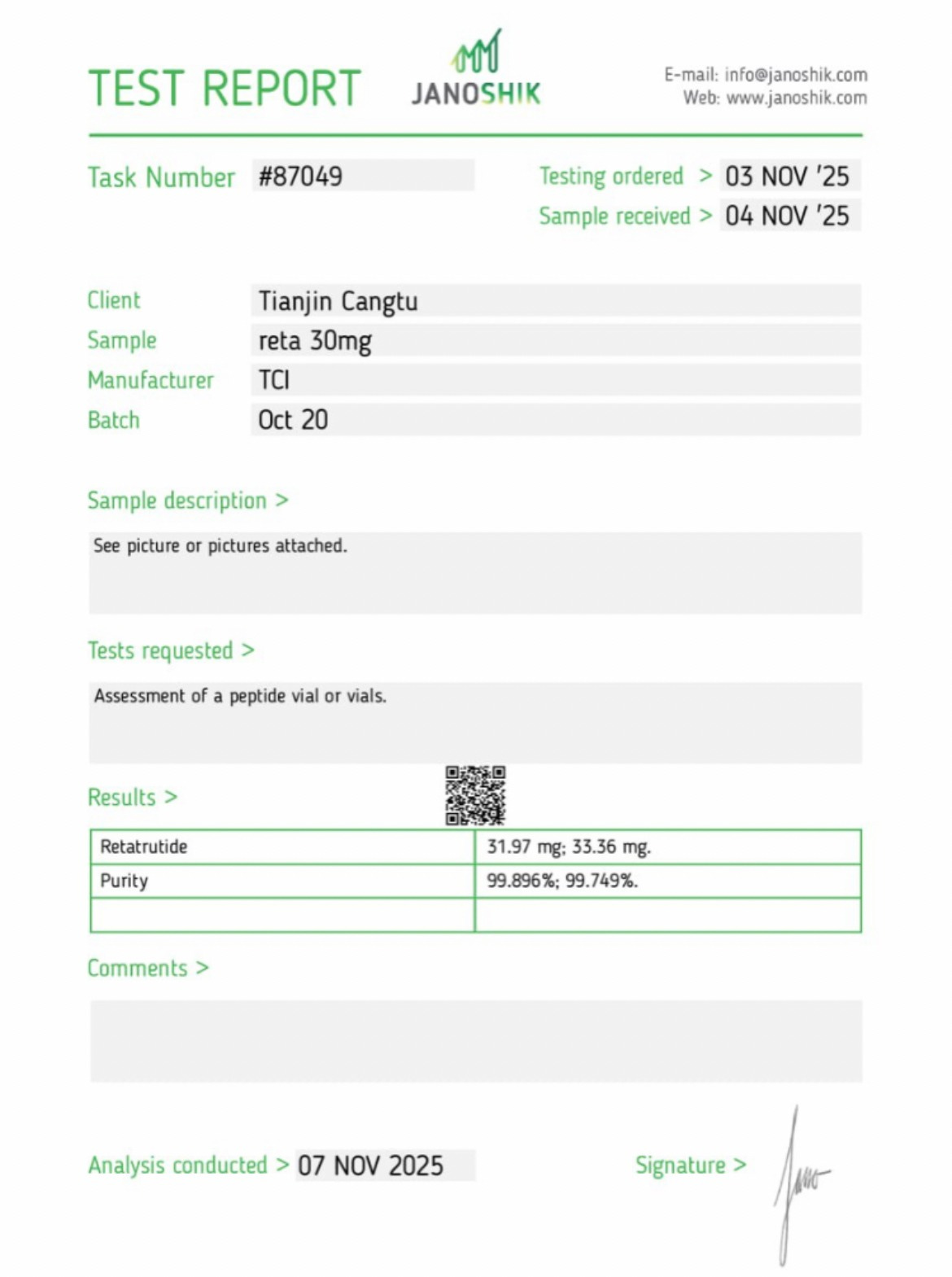
A robust COA typically contains the following:
Batch or lot number tied to the tested product
Testing date
Lab name and accreditation
Analytical methods used
Raw analytical data (chromatograms or spectra)
Quantitative test results
Above, we see many of the indications of a robust COA. Lab name in the upper left corner, testing date in the upper right. A listing of the analytical methods used (RP-HPLC with UV detection) and the raw data in the form of a chromatogram, along with the quantitative results.
Such transparency matters because analytical chemistry is a process with traceable steps. Therefore a trustworthy report ought to show its work.
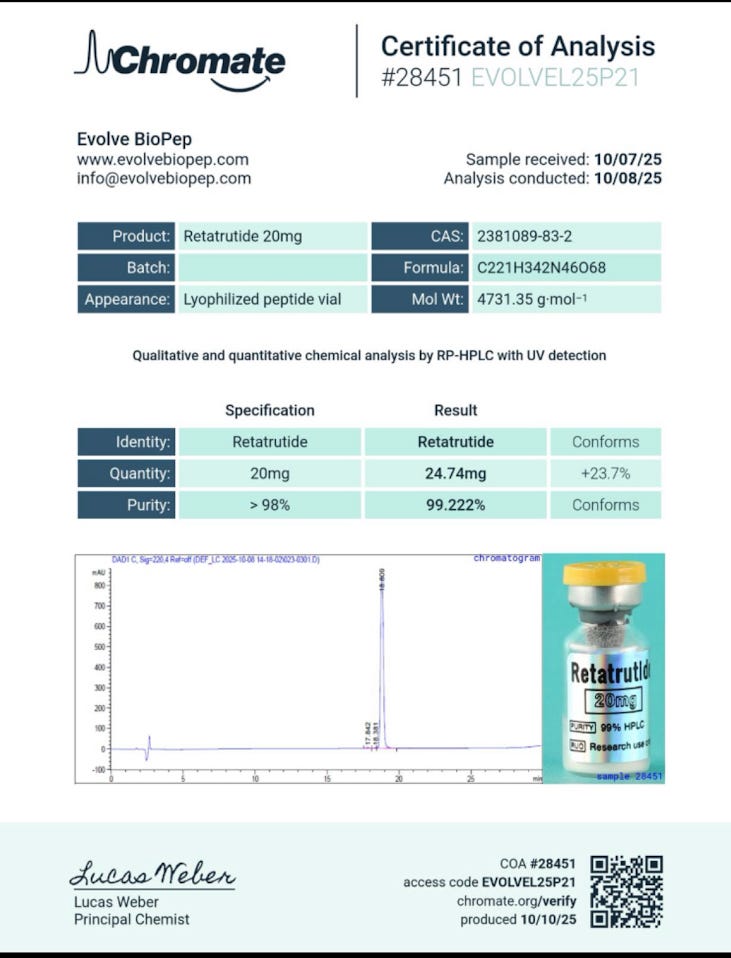
By contrast, a weaker COA might look like this:
No raw analytical data, no real sense of the testing methods, not even a number or QR code to verify the test online. With little tethering this COA to a specific batch of retatrutide, it becomes eminently recyclable. One can imagine the shady group buy organizer terrorizing his customers with such a document, using a little Dunning-Kruger effect arrogance:
Purity: 99% PASS
Such a limp summary of an unknown chemical instills little confidence in the consumer. Imagine a health inspection report that reads: “Food: Probably fine.” You might think twice before eating at that restaurant.
In such cases the COA’s work is less analytical than theatrical. It signals legitimacy without doing the hard work of demonstrating it. Beware the supplier who would show you a report like this. You’re better off – i.e. safer – ordering your amino chains elsewhere.
More on COAs and illusions of trust:






Awesome article! I would like to hear some more information about the amount of excipients, fillers, residual solvent tests, and TFA testing. A 99%+ result from an HPLC doesn't mean 1% of the vial isn't peptide. Its pretty common to see filler/excipient be more than triple the weight of the peptide in the vial. I would love to hear your thoughts and insights on all this. Cheers!